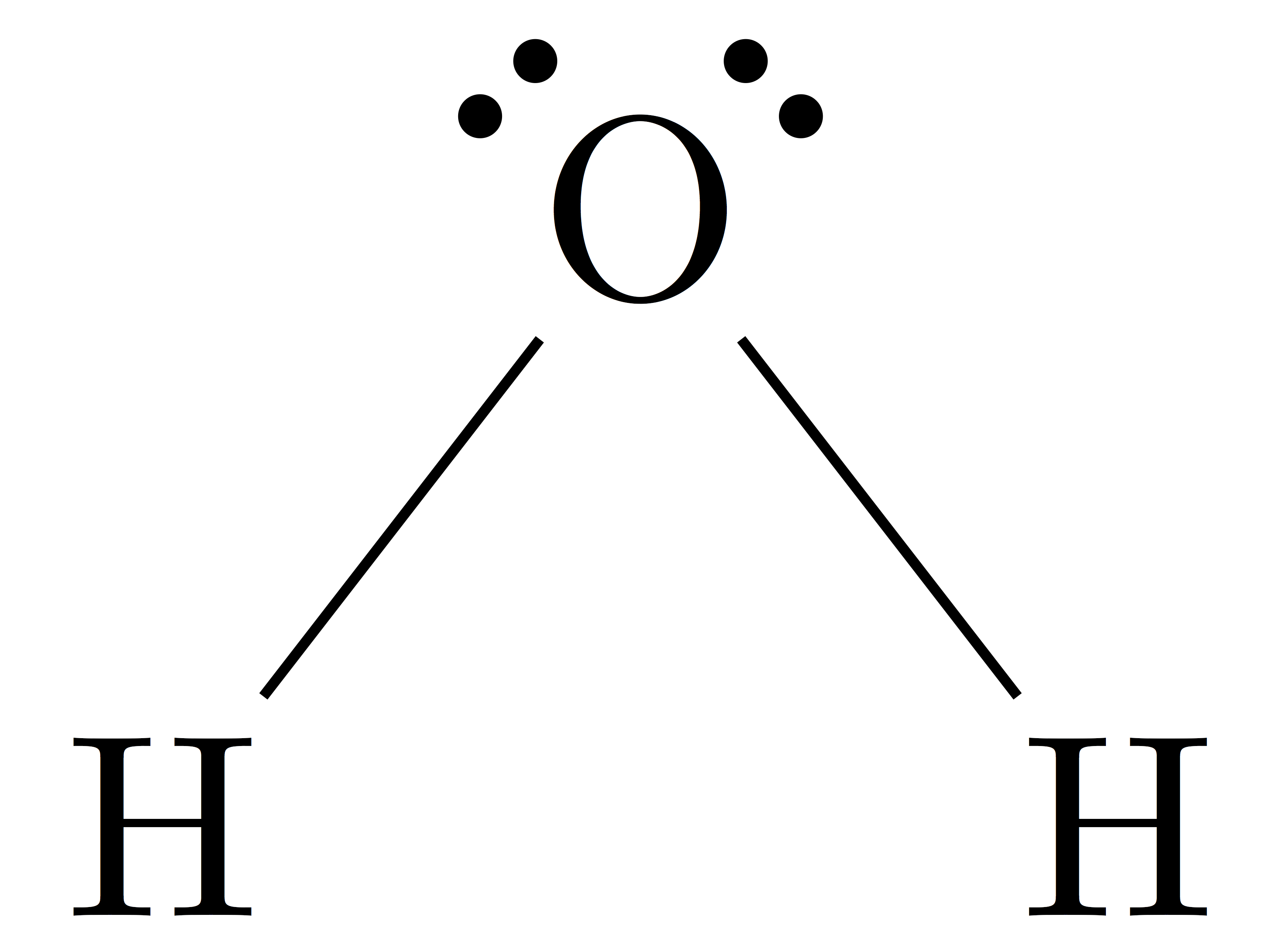
Since it is a theory, it has some drawback, most notably in the prediction of para/diamagnetic behaviour of molecules. The shape of the molecule/ ion depends on the number of lone pairs present. Sp 3d square-based pyramidal (d z 2- y 2 orbital takes part in it) Sp 3d trigonal bipyramidal (dz 2 orbital takes part in it) A sigma bond is formed by front-on overlap of atomic orbitals while pi bonds are formed by side on overlap.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
